One of the easiest mistakes in biosimilar strategy is assuming the formulary decision is the hard part. More often, the challenge starts after the policy is set.
Pharmacy leaders know how to do the biosimilar strategy work upfront. They evaluate the market, build coverage rules, update formularies, and negotiate around cost. But none of that guarantees success once the decision moves into live operations. At that point, the real test begins. Providers have to prescribe through the change, members have to navigate it without disruption, and internal teams have to keep the process moving without adding friction.
That is where a biosimilar strategy can start to lose momentum.
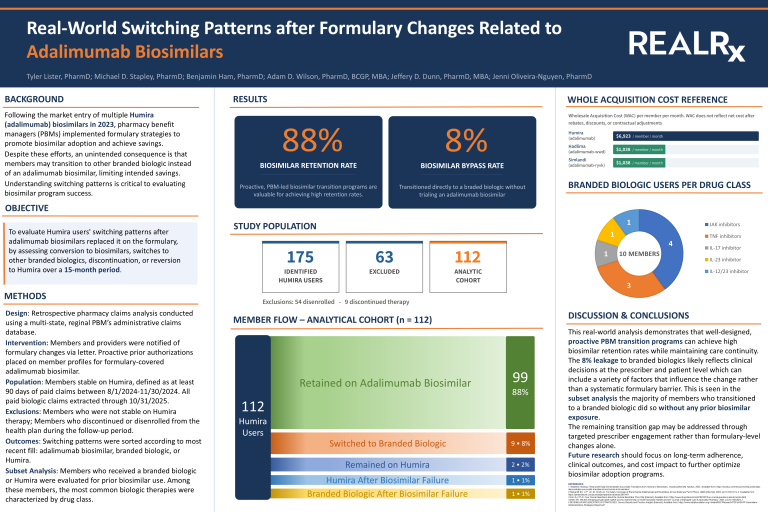
NeosRx clinical pharmacist Tyler Lister, PharmD, presented a poster at the Academy of Managed Care Pharmacy (AMCP) 2026 Annual Meeting: Real-World Switching Patterns after Formulary Changes Related to Adalimumab Biosimilars. This real-world analysis offers a useful example of the realities of biosimilar transition. In the claims analysis of a sample group of stable Humira users, 88% were ultimately retained on an adalimumab biosimilar after the formulary change. Only 8% moved directly to another branded biologic without first trying a biosimilar. The numbers matter not just because they show high biosimilar retention but because of the intentional transition approach that created these results.
Biosimilar strategy is often discussed at the market level: pricing pressure, rebate dynamics, formulary coverage, and product competition. While important, these do not carry a strategy through implementation. A preferred biosimilar on paper still has to hold up once it reaches prescribing behavior, member questions, prior authorization workflows, and the operational friction that comes up after implementation.
The NeosRx clinical team’s study findings also show how the transition was managed in practice. Members and providers were notified of the formulary change, and proactive prior authorizations were placed on member profiles for adalimumab biosimilars. Those details may not grab attention at first glance, but the operational details are often what determines whether a strategy translates into actual change. When the preferred path is easier to follow, adoption is more likely to occur.
Anyone who has worked in pharmacy benefit operations has seen the opposite happen. A strategy can make sense in the contract and still lose traction in the field. The policy is sound, but the rollout is uneven.
Biosimilar adoption fits that pattern. It is partly a formulary issue, but it is also an execution issue. Providers need to understand what is changing and why. Members need a transition they can get through without unnecessary disruption. Clinical teams need processes that support continuity instead of creating more cleanup work later.
The study findings are especially useful when looking at the members who did not follow the intended path. Among the 112 members analyzed, 8% bypassed an adalimumab biosimilar and moved directly to a branded biologic. Tyler Lister and his team concluded that this likely reflects prescriber- and patient-level decisions rather than a broad formulary barrier. It also noted that many members who moved to a branded biologic did so without prior biosimilar exposure. That suggests that, in at least some cases, the next opportunity may sit with provider engagement and transition support, not just tighter formulary restrictions.
The broader relevance of this study goes beyond adalimumab. Specialty pharmacy is full of strategies that appear strong during design and much less convincing during operations. The same pattern shows up in prior authorization, channel management, site-of-care changes, and formulary updates more broadly. The gap is rarely analytical. More often, it is operational.
What this study shows is not simply that high biosimilar retention is possible. It shows that retention is more likely when the transition is actively managed. A proactive, PBM-led approach can preserve continuity, reduce friction, and keep the formulary strategy intact as it moves into real-world practice.
Provider communication matters. Member support matters. Workflow design matters. In biosimilars, as in so many areas of managed care, execution is not separate from the strategy. It is the strategy.